Input the molecular formula and the weight is calculated. We all know that a compound is formed by combining two or more elements metal or nonmetal in a specific ratio by weight.

Nitrogen Element Discovery Production Uses And Fixation Nitrogen Molecules Element Project
Benzene Gas - Specific Heat vs.

. Another feature of molecular sieve adsorbents is their ability to separate gases or liquids by molecular size or polarity. Data should be separated in coma space tab or in separated lines. Air - Molecular Weight and Composition - Dry air is a mixture of gases where the average molecular weight or molar mass can be calculated by adding the weight of each component.
An organic compound possesses a C-H bond while an inorganic compound lacks the same. Thus Mgaseous nitrogen 280134. Note that rounding errors may occur so always check the results.
1 grams Nitrogen is equal to 0071394404106606 mole. Calculate the molecular weight of a formula. Urea nitrogen Unit Conversion between mgdL and mmolL.
Also provides the atomic number element name base atomic weight and formula weight for the calcutated molecular formula. Air - Molecular Weight and Composition - Dry air is a mixture of gases where the average molecular weight or molar mass. Nitrates and ammonia dissolve.
Nitrogen I oxide solubility is 12 gL and nitriloacetate salt solubility is 640 gL whereas nitrogen chloride is water insoluble. Fill in one box to get results in the other box by clicking Calculate button. Figures and tables showing density and specific weight of nitrogen N 2 at temperatures ranging from -175 to 1325 C -280 to 2400 F at atmospheric and higher pressure - Imperial and SI Units.
As a result all four electrons contained in the atomic orbitals in the outermost shell of the nitrogen atom can participate in hybridization making it SP3. Nitrogen - Prandtl number vs. For example water has a.
Nitrogen N 2 solubility at 20 o C and pressure 1 bar is approximately 20 mgL. Molecular weight of Nitrogen or mol The molecular formula for Nitrogen is N. Temperature - Specific heat of Benzene Gas - C6H6 - at temperatures ranging 250 - 900 K.
In other cases ionic compounds have. For example the molecular weight of nitrogen is Mnitrogen 140067 gmol. Molecular sieves will therefore remove gas or liquid impurities to very low levels ppm or less.
The SI base unit for amount of substance is the mole. All plants utilize nitrogen N in the form of NO3- and NH4. It is most imperative element for proper growth and development of plants which significantly increases and enhances the yield and its.
The NH4 ion has no pi bonds. Solubility of nitrogen and nitrogen compounds. According to Merriam-Webster and the Online Etymology Dictionary the word molecule derives from the Latin moles or small unit of massThe word is derived from French molécule 1678 from New Latin molecula diminutive of Latin moles mass barrier.
Molecular masses are calculated from the atomic masses of each nuclide present in the molecule while relative molecular masses are calculated from the standard atomic weights of each elementThe standard atomic weight takes into account the isotopic distribution of the element in a given sample usually assumed to be normal. Molecular weight M is defined as the number of grams g per mol of a substance. Use this page to learn how to convert between grams Nitrogen and mole.
The pore or cage openings are the same size as many molecules eg in the case of hydrocarbon paraffins straight. M is obtained from standard periodic charts or periodic tables of the elements. There are various types of compounds which are categorized according to the constituent ingredients.
The word which until the late 18th century was used only in Latin form became popular after being used. Nitrogen solubility may differ between compounds. Nitrogen in its gaseous or vapor state occurs as a diatomic molecule N2.
In NH4 nitrogen and the 4 hydrogen atoms make 4 sigma bonds out of which 3 are covalent bonds and the fourth one is a dative bond.

Periodic Table Periodic Table Education For All Atomic Number

Nitrogen Chemical Properties Uses Atomic Number Periodic Table Chemistry Basics Chemistry Nitrogen

Nitrogen Definition Symbol Uses Properties Atomic Number Facts Nitrogen Electron Configuration Hydrogen Bond
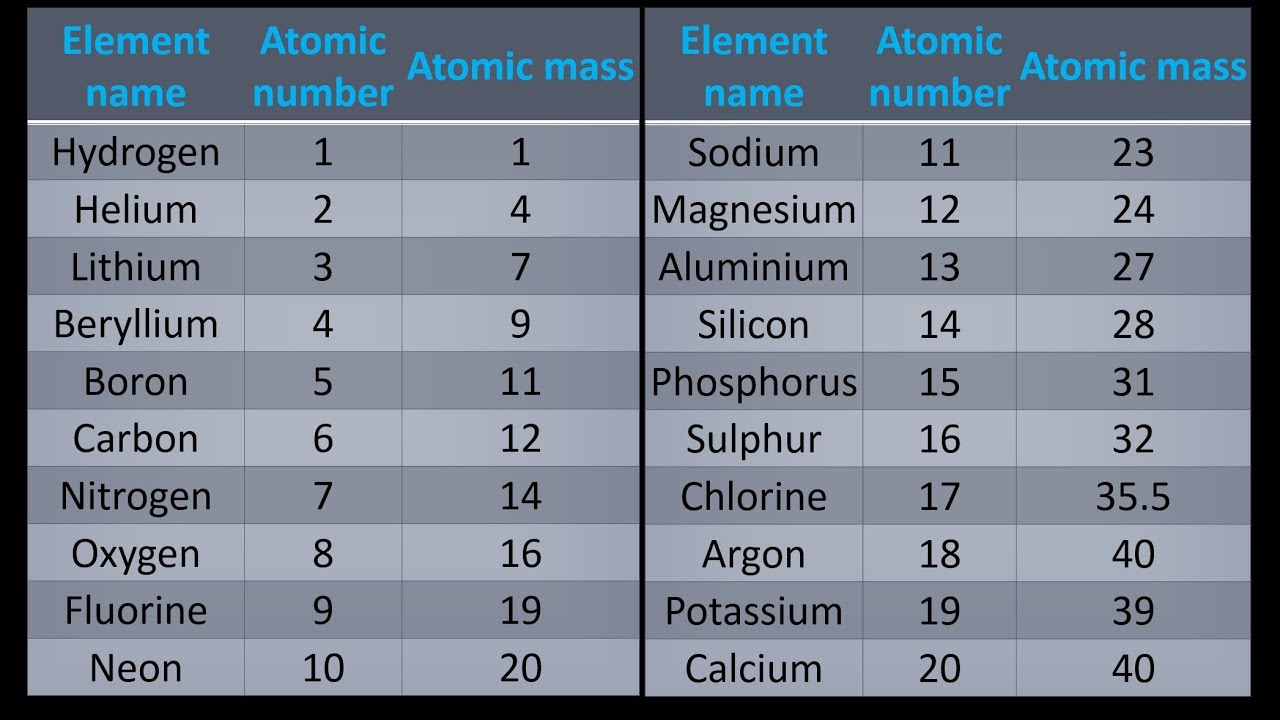
A Simple Way To Get Atomic Mass Of First 20 Elements Of The Periodic Table Youtube Chemistry Lessons Element Chemistry Chemistry Basics